Cell and gene therapies are innovative treatment methods that enable curative approaches for severe, previously incurable diseases. In addition to hematopoietic stem cell transplants, therapies with genetically modified cells have also been approved in the United States and Europe in recent years as advanced therapy medicinal products (ATMPs). These CAR-T cell therapies, in which the patient's own T cells are modified with chimeric antigen receptors (CAR), have so far been used almost exclusively in cancer therapy.
CAR-T cell therapy
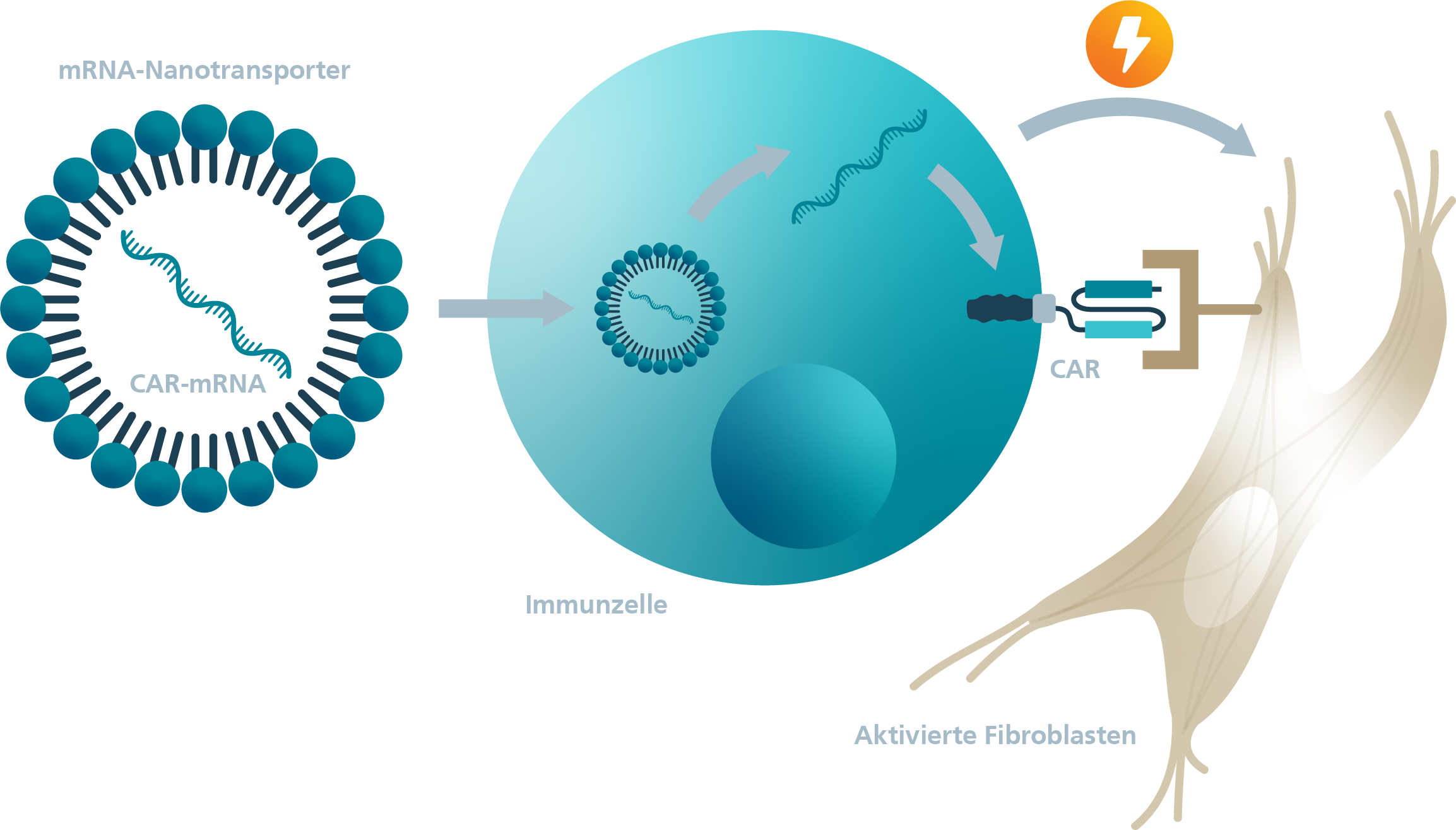
Approved CAR-T cell therapeutics, like the vast majority of new CAR-T cells in development, are based on the stable genetic modification of a patient's own cells using viral vectors. Since CAR-T cell therapy is a very young method, the long-term consequences of off-target effects, for example, have not been sufficiently investigated. Furthermore, persistent CAR-T cells can cause severe side effects. An alternative to stable modification is the temporary modification of cells using a messenger RNA (mRNA) that codes for the CAR protein.
The transfer of the CAR-T cell therapy approach to infectious diseases, autoimmune diseases and fibrosis is the current status of research. In fibrotic diseases, the stroma-immune cell axis plays a crucial role. Therefore, activated, misdirected fibroblasts represent an important target for therapies. In preclinical studies, stably generated CAR-T cells against activated fibroblasts have been successfully used to reduce fibrosis.
 Fraunhofer Cluster of Excellence Immune-Mediated Diseases
Fraunhofer Cluster of Excellence Immune-Mediated Diseases