Optimized implementation of (collaboration) projects
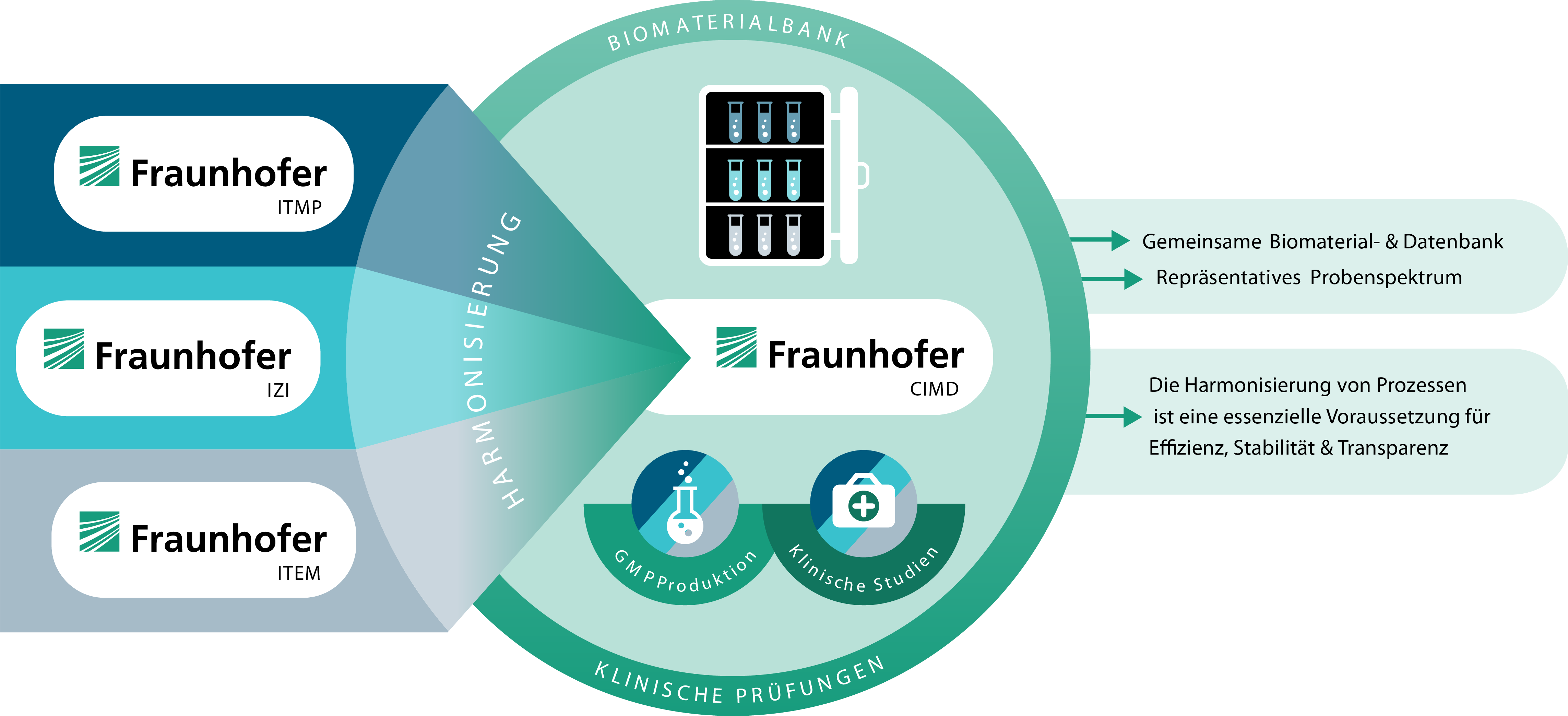
Each core institute has an institute branch, project group or several working groups that have been dealing intensively with various clinical research questions or commissions for years. The aim of this project is to simplify the implementation of joint projects and industrial collaborations in the clinical research section of Fraunhofer CIMD. To this end, a structure is being established that simplifies collaboration between the core institutes and with external partners.
Our goal is to carry out projects in a controlled and standardized manner while creating synergies. To achieve this, we must first select suitable systems that ensure consistent quality at all participating core institutes. To do this, we must first identify systems that, when established and harmonized with the respective processes, represent an increase in quality and create synergies.
The expertise of the institutes in conducting projects in the highly regulated section of Good X Practice (GxP), such as Good Clinical Practice (GCP), Good Manufacturing Practice (GMP) and Good Laboratory Practice (GLP), is used in this project. All institutes base their actions on these internationally applied guidelines. This makes it possible to assess the introduction and the properties of a new electronic system for the standardized, quality-controlled and quality-promoting implementation of (study) projects based on the institutes' many years of expertise.
Outlook
As part of the project, systems such as electronic data capture systems or systems for controlling quality documents are analyzed and possibilities for harmonization are identified. The systems should enable standardized, quality-controlled workflows, as well as simplify and optimize processes. This will allow synergies to be realized and enhance the quality of the partner institutes' projects.
Participating institutes
Fraunhofer Institute for Translational Medicine and Pharmacology ITMP
Fraunhofer Institute for Cell Therapy and Immunology IZI
Fraunhofer Institute for Toxicology and Experimental Medicine ITEM
Project manager:
Dr. Tanja Roßmanith (project manager), Fraunhofer ITMP
Core team structural harmonization of the biomaterial banks
Fraunhofer ITEM: Prof. Jens Hohlfeld, Dr. Philipp Badorrek
Fraunhofer IZI: PD Dr. Stephan Fricke, Dr. André-René Blaudszun, Mirko Müller
 Fraunhofer Cluster of Excellence Immune-Mediated Diseases
Fraunhofer Cluster of Excellence Immune-Mediated Diseases