Joint Lab: Fraunhofer IZI and Cytiva
GMP process development of novel gene transfer methods for cell and gene therapy
Autoimmune diseases comprise a broad spectrum of more than 80 diseases characterized by complex pathologies. These diseases arise when the body's own immune cells, in particular autoreactive B and T cells, trigger irregular attacks on the body's own tissue through various (effector) mechanisms. Among the most common autoimmune diseases are rheumatoid arthritis with about 40 cases per 100,000 people per year and systemic lupus erythematosus (SLE) with 5-10 new cases per 100,000 people per year.
Due to the often quite limited understanding and/or complexity of the underlying molecular mechanisms, current standard approaches to treating diseases such as systemic lupus erythematosus, multiple sclerosis, type 1 diabetes mellitus, psoriatic arthritis and rheumatoid arthritis usually focus on managing the disease rather than curing it.
The main concept for treating autoimmune disorders remains the prevention of autoreactive immune cells from attacking host tissues, by broad immunosuppression, predominantly glucocorticoids and nonsteroidal anti-inflammatory drugs. Additionally, harmful immune cells can be blocked more specifically by therapeutic antibodies. Another, more drastic option is to “reset” the patient's immune system and transplant a “new” foreign immune system through hematopoietic cell transplantation.
Development of new gene transfer methods for cell and gene therapy production
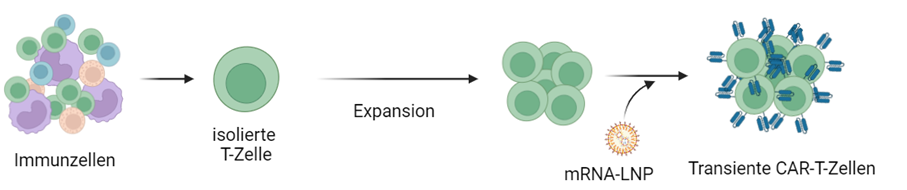
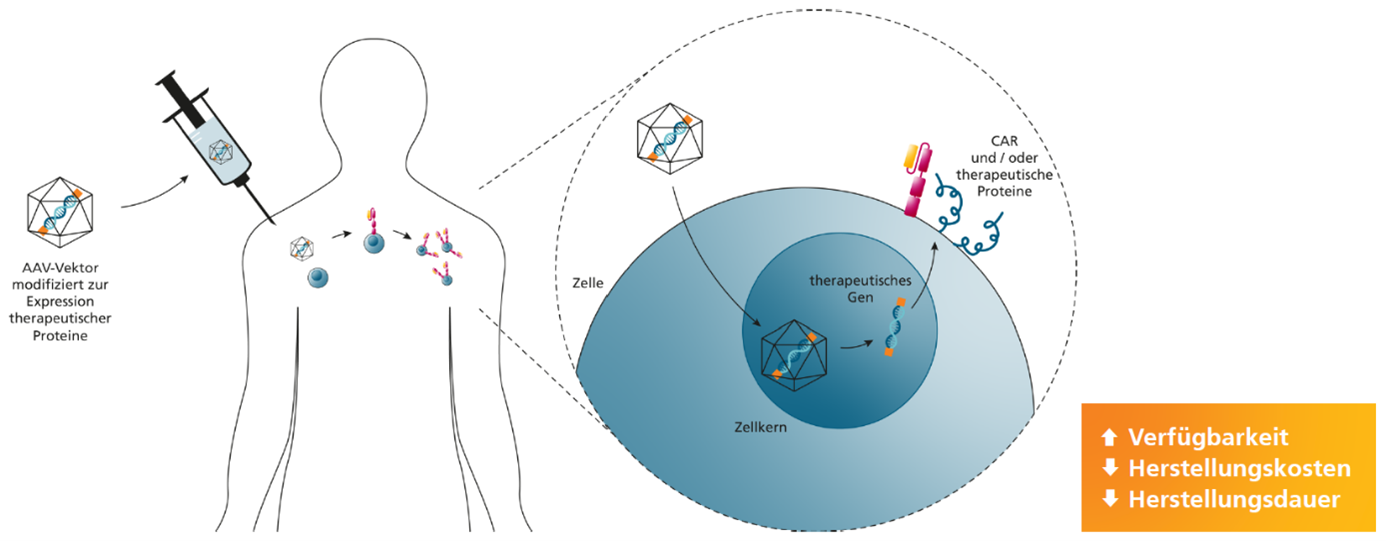
The overarching goal of the project is to develop GMP-compliant methods for novel gene transfer methods for cell and gene therapy production. The process development and optimization will be carried out for the production of adeno-associated virus (AAV) vectors as gene shuttles and for mRNA lipid nanoparticle (mRNA-LNP)-based cell therapies.
The project is being carried out in collaboration with the company Cytiva. The advantage of this collaboration is the possibility to use the existing technical resources and process know-how at Cytiva's locations and combine them with the existing scientific and process engineering know-how at Fraunhofer IZI.
Leading research institute in the sections Cell Therapy and Immunology
Fraunhofer IZI in Leipzig is a leading research institute in the sections Cell Therapy and Immunology. The GMP manufacturing of innovative cell therapies, such as CAR-T cells for clinical application, has been established at the Fraunhofer IZI for many years. In addition, a separate department for cell and gene therapy development with 50 employees has been established to close the value chain from research to clinical application of novel therapies.
 Fraunhofer Cluster of Excellence Immune-Mediated Diseases
Fraunhofer Cluster of Excellence Immune-Mediated Diseases